#NIH clinical trial of investigational vaccine for #COVID_19 begins
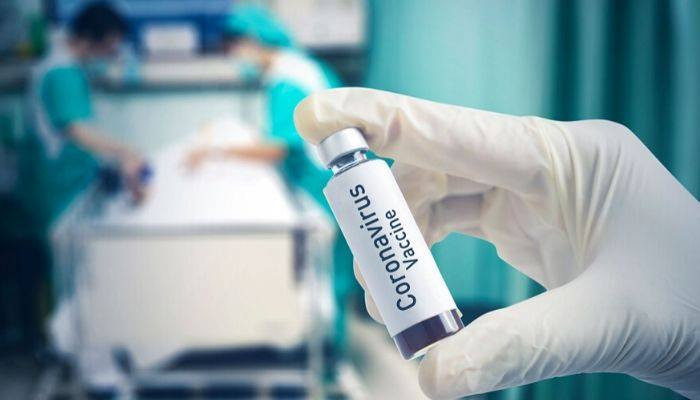 A Phase 1 clinical trial evaluating an investigational vaccine designed to protect against coronavirus disease 2019 (COVID-19) has begun at Kaiser Permanente Washington Health Research Institute (KPWHRI) in Seattle. The National Institute of Allergy and Infectious Diseases (NIAID), part of the National Institutes of Health, is funding the trial. KPWHRI is part of NIAID’s Infectious Diseases Clinical Research Consortium. The open-label trial will enroll 45 healthy adult volunteers ages 18 to 55 years over approximately 6 weeks. The first participant received the investigational vaccine today. The study is evaluating different doses of the experimental vaccine for safety and its ability to induce an immune response in participants. This is the first of multiple steps in the clinical trial process for evaluating the potential benefit of the vaccine. |

Pompeii escape: AI reconstructs the last gesture of an eruption victim (photo)
497Yesterday, 00:37
Mount Erebus blows USD 6,000 worth of gold into the air every day (video)
96906.05.2026, 00:00
They Found a 1,000-Year-Old Treasure Buried Underground (photo)
97203.05.2026, 20:56
Trump promises to release 'pretty interesting' secret UFO files
99829.04.2026, 23:52
«We are pleased to announce the launch of a new international journal entitled «Etiuni–Urartu: Journal of Ancient Armenian Studies»»: Miqayel Badalyan
50327.04.2026, 01:09
Mystery of Noah’s Ark site deepens as experts ‘find underground corridors’ (photo, video)
77726.04.2026, 15:02
